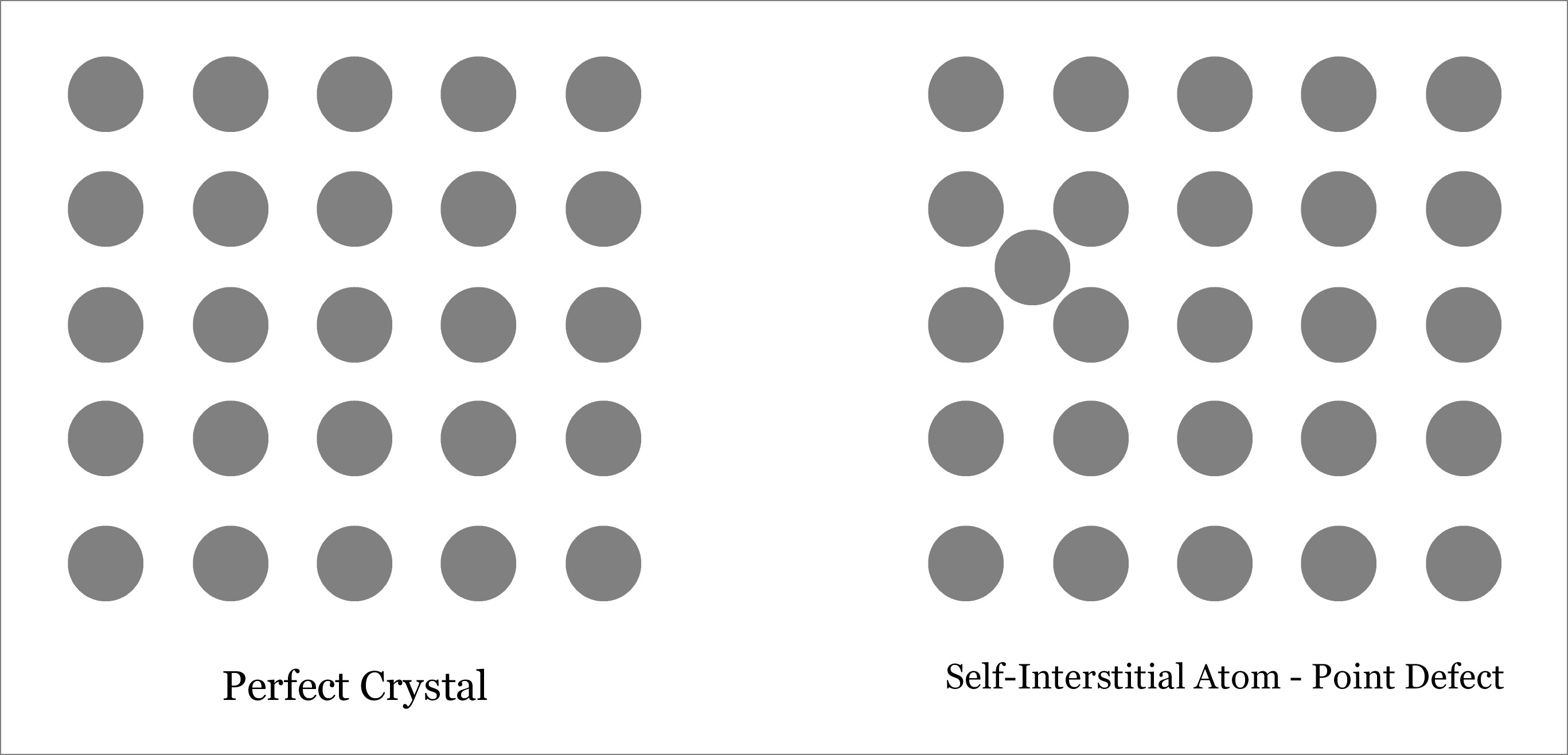
An interstitial defect occurs when an atom takes the interstitial position of the lattice structure. This interstitial atom may be of the same crystal or of a foreign material. Accordingly, interstitial defect can be of two types:
- Self-Interstitial Defect—occurs when atom of the same crystalline solid occupies the interstitial position leaving its original lattice site.
- Interstitial Defect—occurs when a foreign atom occupies the interstitial position.
Although extra atom occupies the empty interstitial space, the size of the atom is usually larger than that of the empty space. Thus the surrounding atoms are compressed and distorted. Presence of substantial number of interstitial atoms can change the mechanical and thermal properties of the solid. However, this is sometime beneficial, and thus interstitial defects can be applied in a controlled way to enhance various properties of the solid. For example, in steel production, carbon is added with iron. Learn more about interstitial defect.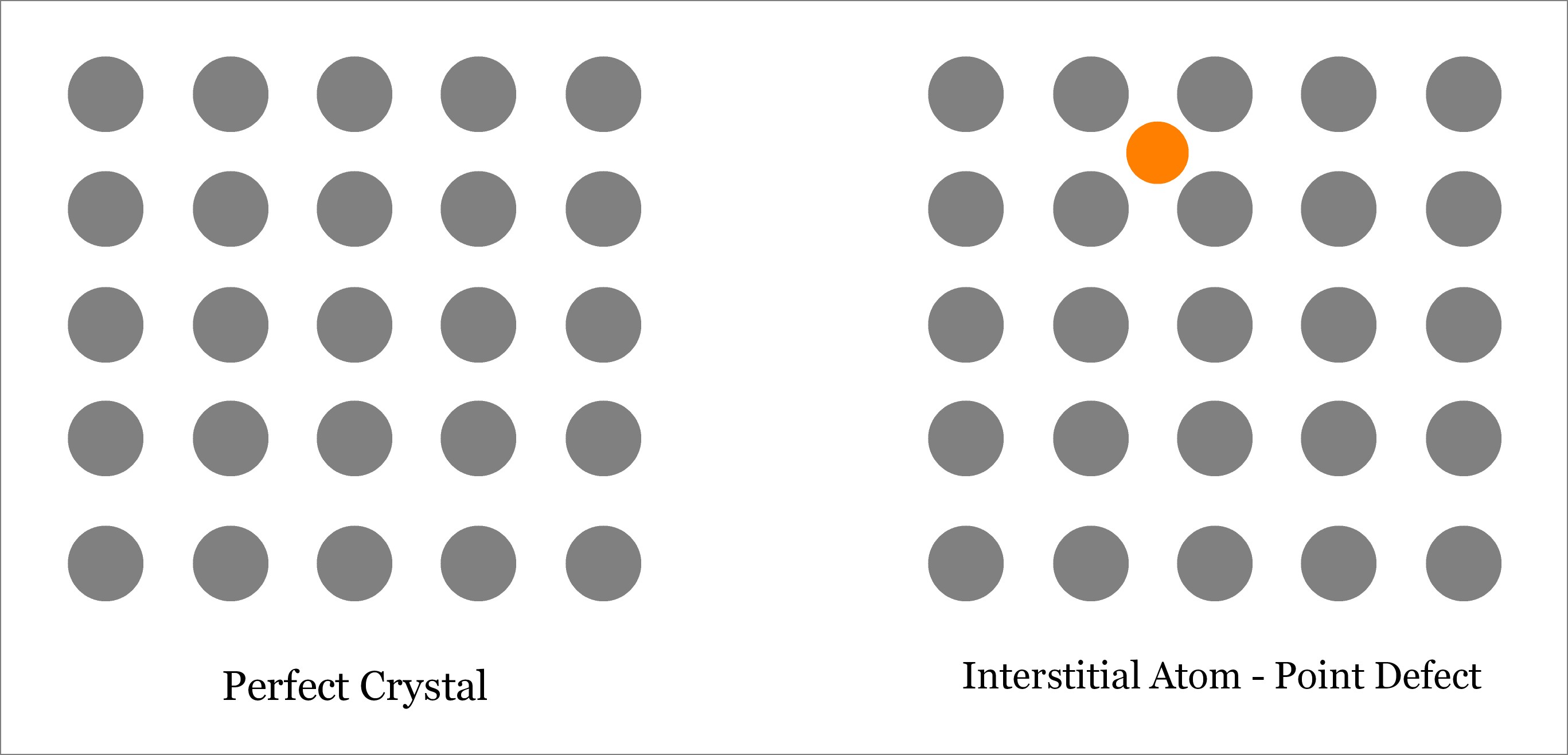
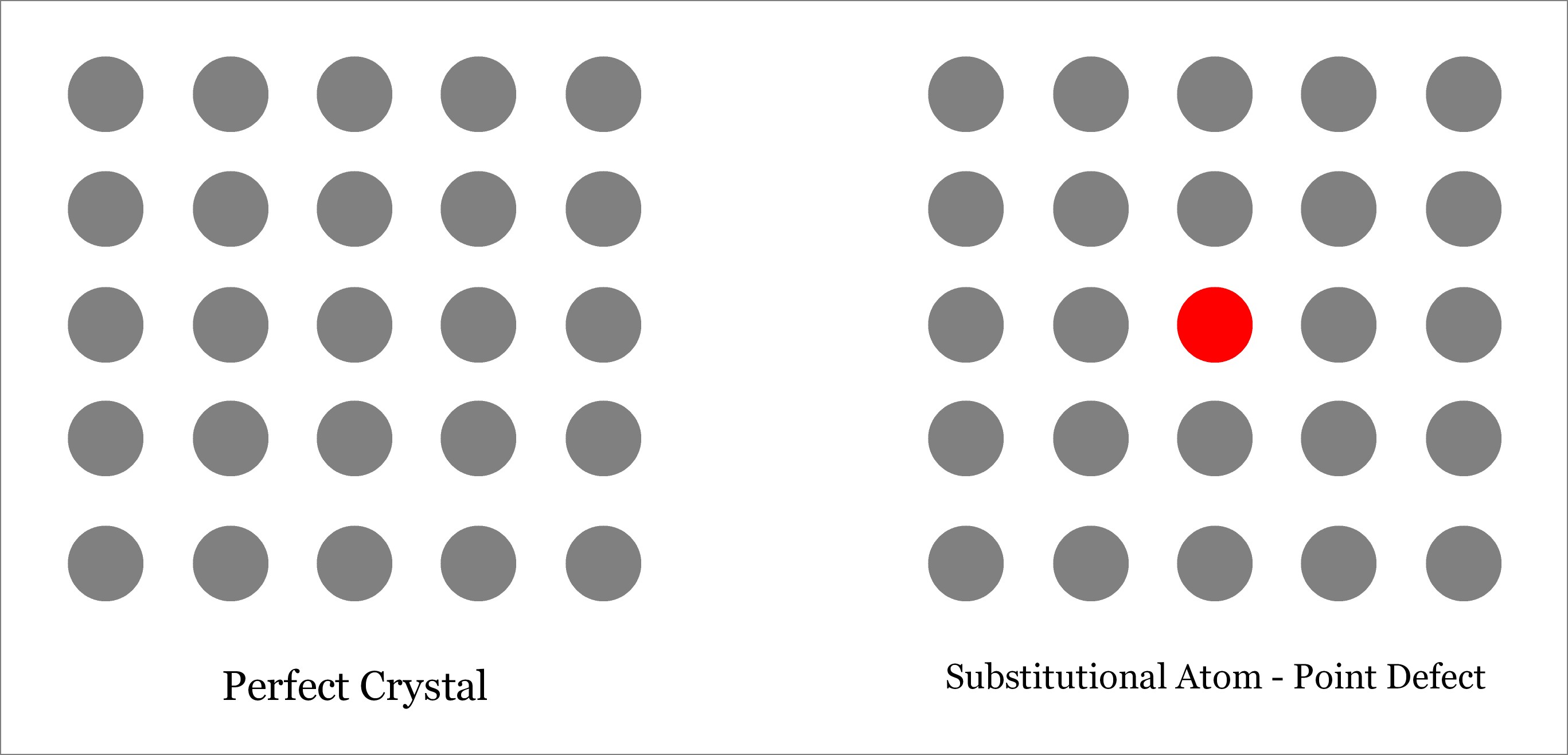
Substitutional Defect occurs when the original atom in the lattice site of a crystalline solid is replaced by a different type of atom. Unlike interstitial defect, foreign atom should occupy the lattice site only and not the interstitial position, as depicted below. The foreign atom may be of same size or different (either larger or smaller). Depending on the size of the substituted foreign atom, the neighboring atoms may remain either in tension or in compression. Learn more about substitutional defect.
Although both are point defects, their mechanisms are different. Difference between interstitial defect and Substitutional defect is tabulated below.
| Interstitial Defect | Substitutional Defect |
|---|---|
| In Interstitial defect, atom occupies the interstitial space of the lattice structure of solid crystal. | In Substitutional defect, the foreign atom occupies only the lattice site of a crystalline solid. |
| Interstitial defect may occur by the atoms of either same solid (called self-interstitial) or foreign material. | Substitutional defect is always caused by foreign atoms. |
| Usually the interstitial atom is larger in size as compared to the space available in the interstitial position, so the neighboring atoms remain under compression. | Substitutional atom can be either larger or smaller as compared to the original atom of the solid crystal. So the neighboring atoms can be either under tension (when foreign atom is smaller) or under compression (when foreign atom is larger). |
| In case of interstitial or self-interstitial defect, the weight and density of the crystalline solid increase due to presence of extra atom. | Although Substitutional defect is caused by foreign atoms, the weight and density of the crystalline solid remain more or less constant. |
References
- Book: Callister’S Materials Science and Engineering by R. Balasubramaniam (Wiley India Private Limited). Buy this book
- Book: Materials Science and Engineering by V. Raghavan (PHI Learning Private Limited). Buy this book
- Book: Materials Science and Engineering: Problems with Solutions by M. N. Shetty (PHI Learning Private Limited). Buy this book